Preview this course
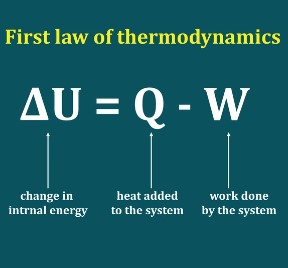
First Law of Thermodynamics in Closed System by PK NAG (Chapter 04)
32 already enrolled!
FREE
32 already enrolled!
Why enroll
Course details
Course suitable for
Key topics covered
Course content
The course is readily available, allowing learners to start and complete it at their own pace.

