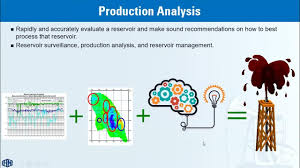
Thermodynamics of Hydrogen Energy Storage
FREE
6 already enrolled!
Why enroll
Course content
The course is readily available, allowing learners to start and complete it at their own pace.
Course details
Course suitable for
Our Alumni Work At
Why people choose EveryEng
Industry-aligned courses, expert training, hands-on learning, recognized certifications, and job opportunities—all in a flexible and supportive environment.
- Industry Veteran
- Trainer Review

KPMG
From learning new technologies to getting hired, this platform changed engineer's career trajectory! It's a great work by Engineer's behind the EveryEng brain.

Worley
I’ve had an amazing experience with EveryEng! The courses are well-structured, insightful, and practical. The platform is perfect for anyone looking to advance their engineering career with industry-relevant skills.

Servilink Systems
EveryEng offers a fantastic learning experience with a great selection of courses and expert mentors. The platform is user-friendly, and the knowledge gained is highly practical. It's been a great journey so far!

SLB
I never imagined an online learning platform could be this effective! EveryEng’s courses are top-notch, the mentors are industry experts, and the skills my team gained have made a real difference in the performance!"

Heelium
EveryEng is my go-to platform for engineering education. The courses are easy to follow, and the instructors are very knowledgeable. The platform has helped my team gain confidence and expertise in our field.
Questions and Answers
No questions yet - Be the first one to ask!