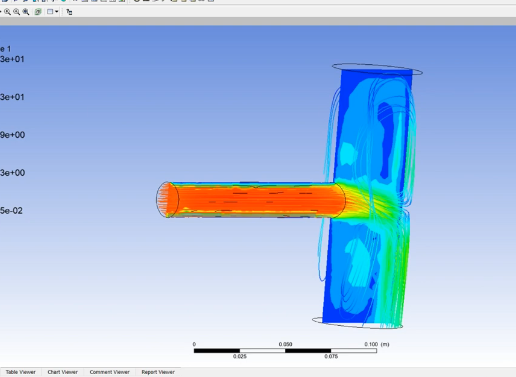
Work & Heat Transfer in Engineering Thermodynamics by PK NAG (Chapter 03)
FREE
103 already enrolled!
Opportunities that awaits you!

Earn a course completion certificate
Add this credential to your LinkedIn profile, resume, or CV. Share it on social media and in your performance review
Course content
The course is readily available, allowing learners to start and complete it at their own pace.
Course details
Course suitable for
Key topics covered
Course Attachments
Our Alumni Work At
Why people choose EveryEng
Industry-aligned courses, expert training, hands-on learning, recognized certifications, and job opportunities—all in a flexible and supportive environment.
- Industry Veteran
- Trainer Review

KPMG
From learning new technologies to getting hired, this platform changed engineer's career trajectory! It's a great work by Engineer's behind the EveryEng brain.

Advanced Upstream
EveryEng helped me bridge the gap between academia and industry. Their practical training and job placement services enabled me to secure a well-paying job and start building a successful engineering career!!

LyondellBasell
The learning experience at EveryEng is truly exceptional! The platform is well-organized, making it easy to find relevant courses. The detailed lessons and expert insights are very helpful and helped greatly to improve engineer's technical knowledge.

Halliburton
I had been looking for a reliable online platform for engineering courses, and EveryEng delivered beyond my expectations. The content is top-notch, the instructors are excellent, and the learning experience is seamless.

Lummus
I love how EveryEng makes complex engineering concepts easy to understand. The platform offers valuable insights, great mentors, and a supportive community. It's truly a one-stop solution for engineers worldwide.
Questions and Answers
No questions yet - Be the first one to ask!