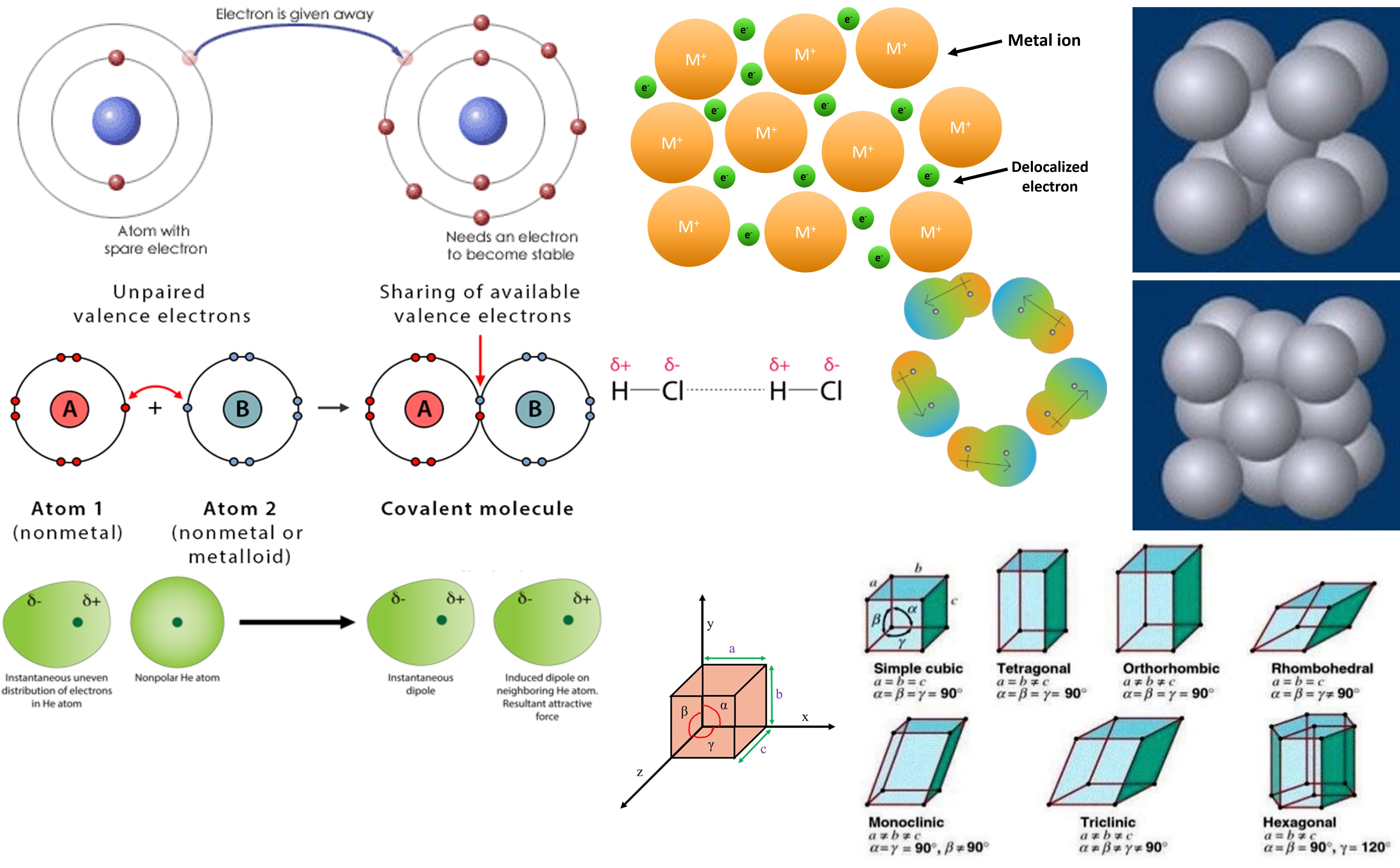
Introduction to Chemical Engineering-II
FREE
3 already enrolled!
Opportunities that awaits you!

Earn a course completion certificate
Add this credential to your LinkedIn profile, resume, or CV. Share it on social media and in your performance review
Course content
The course is readily available, allowing learners to start and complete it at their own pace.
Course details
Course suitable for
Key topics covered
Our Alumni Work At
Why people choose EveryEng
Industry-aligned courses, expert training, hands-on learning, recognized certifications, and job opportunities—all in a flexible and supportive environment.
- Industry Veteran
- Trainer Review

Advanced Upstream
EveryEng helped me bridge the gap between academia and industry. Their practical training and job placement services enabled me to secure a well-paying job and start building a successful engineering career!!

Electric Hydrogen
EveryEng is more than just a training platform - it's a career catalyst! Their expert instructors, real-world projects, and job assistance helped engineers transition into a successful engineering professional.

Worley
I’ve had an amazing experience with EveryEng! The courses are well-structured, insightful, and practical. The platform is perfect for anyone looking to advance their engineering career with industry-relevant skills.

Petrofac
I was skeptical at first, but EveryEng's training programs really delivered. I gained the skills and confidence to take on challenging projects and advance my career. Highly recommended!

SaiDeepa
EveryEng has completely transformed the learning experience. The platform is easy to navigate, offers high-quality content, and provides great support. I highly recommend it to anyone looking to upskill in engineering.
Questions and Answers
No questions yet - Be the first one to ask!